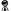The celebrated Iron Pillar of Delhi is widely
regarded an architectural wonder for the way it has defied rust through
the last 1600 years. The Iron Pillar has long been an enigma for
metallurgists who have been unable to unravel the reason behind this
stubborn resistance to corrosion. Made of 98% wrought iron, this tall,
sleek pillar is a bona fide proof of the mastery Indian ironsmiths
achieved in the extraction and processing of iron.
However, in 2002, a report published in the Current Science journal by
experts of IIT, Kanpur offered a scientific explanation to the
rust-resilient nature of the Iron Pillar at Delhi. The scientists, led
by noted metallurgist R. Balasubramaniam, found that the pillar's
anti-corrosion ability stems from the presence of a thin layer of "misawite",
a compound of iron, oxygen and hydrogen. This protective layer took form
within three years after erection of the pillar and has been growing
slowly since.
The film was created by a catalytic reaction triggered off by the high
phosphorous content in the iron-as much as one per cent against less
than 0.05 per cent in today's iron. This substantial presence of
phosphorous content results from the unique iron-making process
practiced by ancient Indians, who reduced iron ore into steel in one
step by mixing it with charcoal.
Read on to know about the corrosion resistance of the Iron Pillar of Delhi.
Colors of India: Iron Pillar : Corrosion
Resistance of the Iron Pillar of Delhi